CD123, a component of interleukin-3 receptor, is overexpressed on the surface of acute myeloid leukemia (AML) blasts and leukemic stem cells compared with normal hematopoietic progenitors, which makes this antigen an attractive therapeutic target. IMGN632 is a novel antibody-drug-conjugate comprising a high affinity CD123-targeting antibody and DNA alkylating cytotoxic payload. In preclinical studies, IMGN632 demonstrated high potency and selective cytotoxicity against AML leukemic cells vs normal myeloid progenitors, suggesting reduced likelihood of myelosuppression (Kovtun, 2018). Importantly, we previously showed high synergy of IMGN632 combination with BCL-2 inhibitor venetoclax (VEN) and DNA hypomethylating azacytidine (AZA) in AML cell lines and xenograft models (ASH 2020, #617). IMGN632 demonstrated encouraging single agent activity and favorable tolerability in relapsed/refractory AML patients (ASH 2019 #734, ASCO 2020 TPS7563, NCT03386513) and is currently tested in combination with VEN and/ or AZA in relapsed and frontline CD123-positive AML patients (NCT04086264). In this study, we provide new insight into molecular mechanisms underlying the synergistic efficacy of these combinations using AML models in vitro.
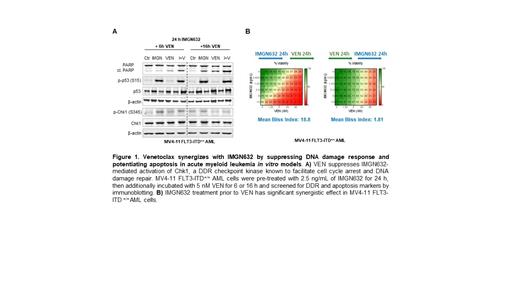
AML cells with FLT3-ITD mutations expressed higher levels of CD123, as previously reported (Han 2013). FLT3-ITD cell lines were particularly sensitive to IMGN632 with IC 50 values at low ng/ml range. Correspondingly, as evidenced by large scale drug combination screen using BLISS independence model, triple IMGN632/VEN/AZA treatment was synergistic at multiple dosage levels and ratios, suggesting a favorable potency of triple combination in FLT3-mutated subtypes of AML associated with poor prognosis. The efficacy of IMGN632/VEN/AZA combination was however decreased in cells lacking functional TP53. shRNA-mediated knockdown of TP53 in FLT3-ITD MOLM13 cells resulted in increased cell viability, reduced apoptosis and downregulation of cleaved caspase-3 and PARP. While both treated wild-type and TP53 KD cells showed robust overexpression of phosphorylated histone H2AX (p-H2AX), TP53 KD cells expressed higher levels of anti-apoptotic MCL-1 and lower levels of pro-apoptotic BAX, indicative of their intrinsic inability to fully execute apoptosis and resulting in suppressed response to triple combination. Importantly, in sensitive AML cells, IMGN632 induced profound DNA damage, G 1 (MOLM13 and MOLM14 cells) or G 2/M (MV4-11 cells) phase cell cycle arrest and decline in S-phase population, all above augmented by VEN/AZA co-treatment. Observed increase in p-H2AX was accompanied by apoptotic caspase-3 and PARP cleavage and was fully prevented by QVD-OPh caspase inhibitor, suggesting that double-stranded DNA damage and p-H2AX triggered by triple combination were secondary events associated with apoptosis but not the initial cause of cell cycle arrest. Thus, given that cytotoxic payload of IMGN632 is a DNA alkylator, we next tested its impact on mediators of the DNA damage response (DDR). IMGN632 alone induced phosphorylation of p53 at Ser15, a step required for p53 transcriptional activity, and triggered activating phosphorylation of checkpoint kinase Chk1 (Ser345), known to facilitate cell cycle arrest and DNA damage repair, thus preventing treated cells from progressing through S-phase but also increasing the potential of repairing IMGN632-induced DNA damage (Fig. 1A). Upon combined IMGN632+VEN treatment, p53 phosphorylation was sustained, but p-ChK1 decreased, suggesting that VEN may impair protective DDR triggered by IMGN632, resulting in greater cell death. To support this hypothesis, we showed that IMGN632 treatment prior to VEN produced significant synergistic activity (Fig. 1B). Together, these results suggest that VEN, apart from its canonical inhibitory effect on anti-apoptotic BCL-2, exerts previously unrecognized ability to suppress DDR program in AML and augments activity of DNA damaging IMGN632. Failure of cells to sustain DDR in the presence of VEN constitutes a key aspect of high efficacy of IMGN/VEN/AZA combination in AML.
Disclosures
Daver:Jazz: Consultancy; Gilead: Consultancy, Research Funding; AROG: Consultancy; Servier: Consultancy, Research Funding; Genentech: Consultancy, Research Funding; Hanmi: Research Funding; FATE: Research Funding; Astellas: Consultancy, Research Funding; Agios: Consultancy; Celgene: Consultancy; Trillium: Consultancy, Research Funding; Glycomimetics: Research Funding; Trovagene: Research Funding; AbbVie: Consultancy, Research Funding; Novartis: Consultancy; Novimmune: Research Funding; ImmunoGen: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding; Bristol-Myers Squibb: Consultancy, Research Funding; Amgen: Consultancy, Research Funding; Daiichi Sankyo: Consultancy, Research Funding; Kite, a Gilead company: Consultancy, Research Funding; Syndax: Consultancy; Shattuck Labs: Consultancy; Kronos Bio: Research Funding. Watkins:Immunogen Inc.: Ended employment in the past 24 months. Sloss:Immunogen Inc.: Current Employment. Zweidler-McKay:Immunogen Inc.: Current Employment. Konopleva:AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Clinical Trials Support, Research Funding.